Interdisciplinary Projects and Clinical Studies
SNIFFPHONE: Smart Phone for Disease Detection from Exhaled Breath
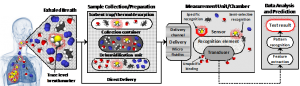
Screening for early detection of a disease is required to reveal groups of individuals from the general population in whom the likelihood of the disease is increased and who could benefit from further medical evaluation. The ideal screening test is high-accuracy, low-cost, non-invasive, easily repeatable, effortlessly operated by a lay-person and has minimal impact on the person’s daily activities. In the SNIFFPHONE project, we aim to tackle these requirements by integrating heterogeneous micro- and nano-technologies into autonomous smart system that can be attached to a mobile phone and analyze disease markers from exhaled breath. In this approach, an interaction between breath sample and a miniaturized array of highly sensitive nanomaterial-based chemical sensors is recorded, stored and pre-processed by integrated miniature on-chip microfluidics and electronics, and then the relevant electrical signals are transferred wirelessly via the mobile phone’s internet to an external server. Statistical pattern recognition methods are then applied on the received data and a clinical report including the screening results is sent back to the designated receiver (e.g., specialist, family doctor) in case of positive result is revealed. SNIFFPHONE represents a new concept addressing major societal challenges in health and well-being of the general population, while taking into account constituent ethical and security aspects. The SNIFFPHONE end-product will integrate functionalities that are relevant to the health screening applications with decreased size, decreased costs, increased predictive and cognitive functions and full autonomy with energy management as well as with operation/use management.
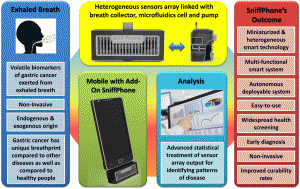
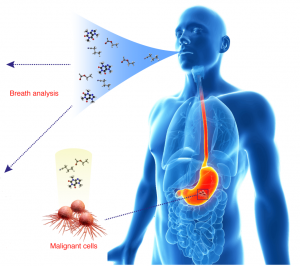
Schematic illustration of the main components and features of the SNIFFPHONE: exhaled breath, heterogeneous sensors array linked with breath collector and microfluidic cell, and advanced statistical and pattern recognition methods. The expected outcome of the SNIFFPHONE’s project is presented as well.
LUMASENSE: A Non-Invasive Test for Guiding Follow–up of Patients with CT-Detected Lung Nodules
This project addresses an urgent clinical need in the field of lung cancer (LC). The rationale behind the project is that CT-based LC screening programs are emerging in many countries worldwide. However, the high false positive rate of these techniques (96% of the 24% positive CT findings are non-cancerous) remains a major challenge. This is due to the fact that non-cancerous CT-detected group has undergone unnecessary invasive procedures that are both costly and associated with significant morbidity and mortality.
This project aims to increase the specificity of the LDCT-based screening program by utilizing an auxiliary noninvasive breath test to distinguish between malignant and non-cancerous CT findings. The lab’s breath test relies on a novel stand-alone device that provides improved speed, sensitivity and portability, as well as simplicity, ease, and low production cost. This project has the potential to immediately become a powerful tool for managing nodule-positive patients. It will both reduce the rate of unnecessary invasive procedures, and prevent treatment delays when cancer exists. This represents a savings of ~$12-16 billion for healthcare systems worldwide.
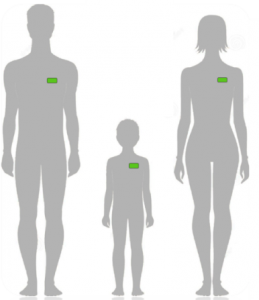
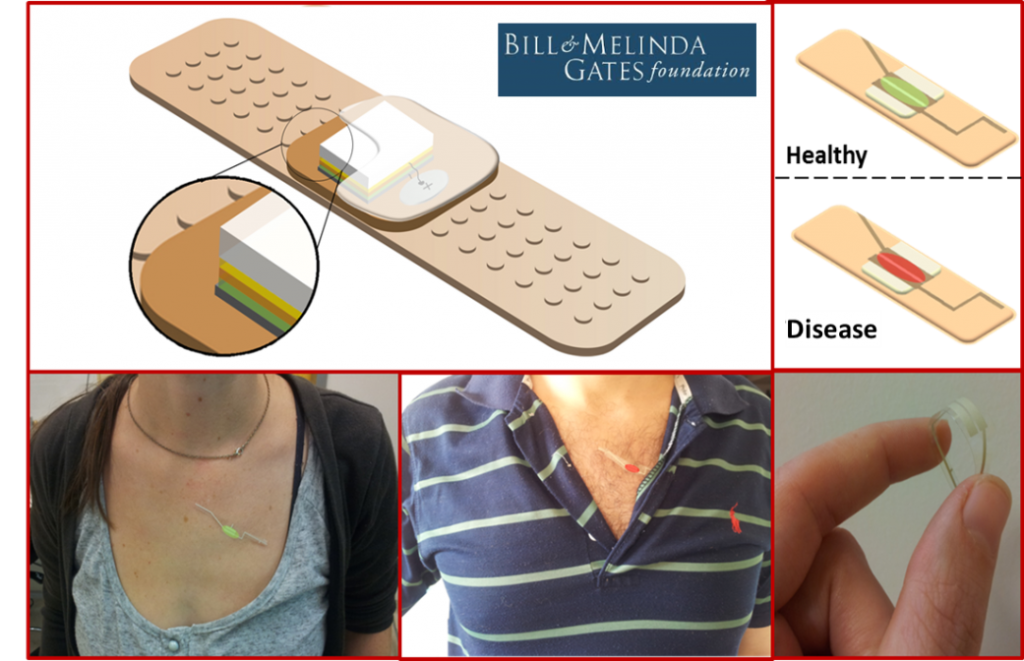
GCE-II: Self-Administered Adhesive Patch for Detection of Tuberculosis - The Bill and Melinda Gates Foundation
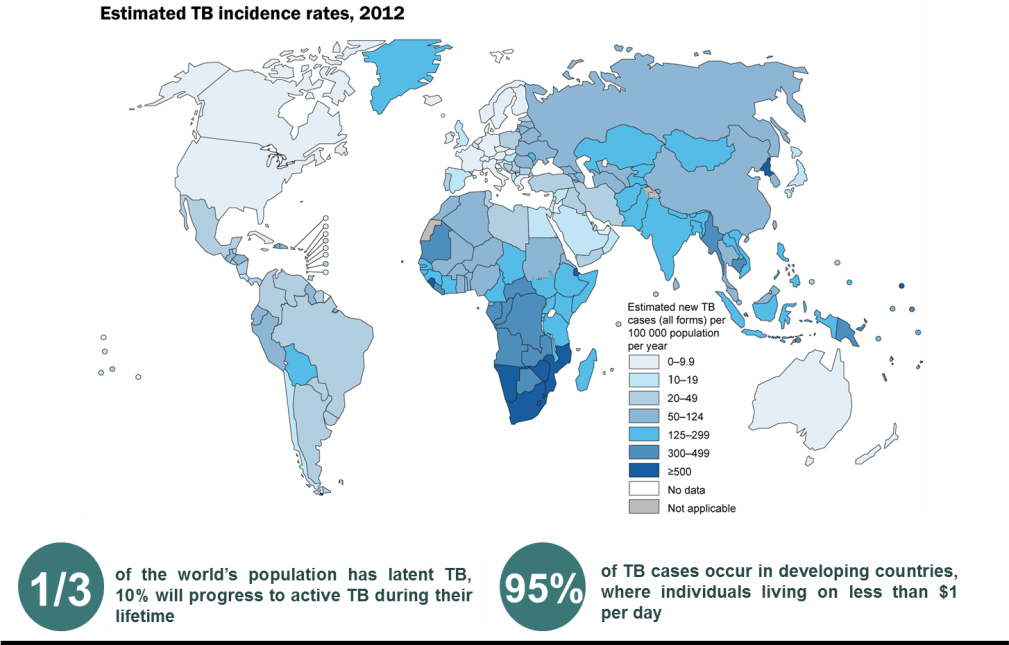
More than two billion people are estimated to be infected with M. tuberculosis (TB), of which 10% progress to active tuberculosis during their lifetimes. About 95% of TB cases occur in developing countries, where individuals live on 1$ per day. This project aims to develop a stand-alone TB test involving an adhesive patch that allows immediate diagnosis of TB from skin samples within two to five minutes, at much lower costs than with current approaches. The novel TB sensing patch is applied on the patient’s chest, either in the clinic or by the patient at home, and will be suitable for children. The possibility of home testing will allow patients to participate actively in the prevention and detection of TB. At-risk populations such as health workers, family members of TB patients, smokers and HIV-carriers will be able to continuously monitor themselves. This two to five minute examination displays a GREEN LIGHT when the user is healthy (i.e., has no TB) or a RED LIGHT when active TB is present.
The development of rapid, accurate, and affordable sensing patches for diagnosis of ‘active TB pulmonary disease vs. non-TB pulmonary disease and latent TB’ will contribute to a greater survival rate of TB patients, and, also, in arresting transmission of the disease. Successful completion of these short- and long-term goals will provide a TB sensing patch. This patch could be employed at resource-poor central reference laboratories, hospital and clinic laboratories, and/or at the point-of-care, helping increase TB screening, and saving at least 400,000 lives yearly.
VOLGACORE: Volatile Biomarkers for Early Detection and Characterization of Gastric and Colorectal Neoplasms
Gastrointestinal cancers, in particular gastric (GC) and colorectal cancers (CRC) remain important healthcare issues globally. No non-invasive screening marker is available for GC, and currently available screening markers for CRC demand great improvements. Previous research by LNBD researchers and others suggests that detection of volatile organic components (VOCs) in exhaled breath may be a promising tool for screening for these cancers. Therefore, the objectives of the project are the development and validation of novel, non-invasive, easy to use and low-cost tools for screening, early detection of GC, CRC and the related premalignant lesions. The project brings together the leading centers in nanotechnological breath analysis with clinical, laboratory and epidemiology experts. Recruitment is done in high-risk regions; involvement of an innovation-oriented SME is planned. VOCs are analyzed by highly sensitive, cross-reactive, nanomaterial-based gas sensors that can identify and separate volatile marker patterns. In addition, particular components are identified with gas chromatography combined with mass spectrometry. A characteristic sensor technique by correlation of the results to GC-MS is developed for the targeted cancer types and the related pre-malignant lesions. Chemiresistors are based on cross-reactive, chemically diverse layers of organically stabilized spherical gold nanoparticles. In addition, a lowcost instrument for VOC testing, based on molecule-terminated nanoparticle films have been developed for use in point-of-care testing settings.
The lab addresses other factors on VOCs.. Researchers give particular attention to gastrointestinal microbiota. They analyze microbiota from biopsies by sequencing a hyper-variable region of the 16S rRNA gene, in combination with sample-specific barcode sequences. The lab thoroughly evaluates the implications of these findings for population-based cancer screening programs.
BIRAX: Rapid Diagnostic Test for Parkinson's Disease to Determine Underlying Mechanisms and Possible Regenerative Therapies.
It can take some time for people with Parkinson’s to be diagnosed. One reason for this is that there is no simple test for accurately diagnosing or monitoring Parkinson’s. Earlier-stage diagnosis will result in better patient treatment.
Recently, the LNBD group has developed a breath test that can be used to analyze the molecules linked with Parkinson’s. This breath test can differentiate between people with Parkinson’s and those of a similar age without the condition.
In collaboration with Prof. John Fienberg of the Technion – IIT and Prof. Roger Barker of Cambridge University, the team is working to test this novel method of diagnosing Parkinson’s. LNBD researchers investigate whether the breath test can be used to diagnose and monitor the progression of Parkinson’s over time, by testing it on a larger group of participants. They look for subtle differences between afflicted individuals to gauge if the test is sensitive enough to identify different forms or types the disease.
The lab also analyzes the molecules in breath samples of Parkinson’s patients for new clues of what causes the condition. Researchers hope their findings will contribute to new Parkinson’s treatment.
This research also promises to shed new light on different molecules that play a role in the development of Parkinson’s. This information may well provide scientists with new targets for therapies that could slow or stop the progression of Parkinson’s.
DIAG-CANCER: Diagnosis, Screening and Monitoring of Cancer Diseases via Exhaled Breath Using an Array of Nanosensors
Cancer is rapidly becoming the greatest health hazard of our days. The most widespread cancers are lung cancer (LC), breast cancer (BC), colorectal cancer (CC), and prostate cancer (PC). The impact of various techniques used for diagnosis, screening and monitoring these cancers is either uncertain and/or inconvenient for patients.
This project aims to create a low-cost, easy-to-use and non-invasive screening method for LC, BC, CC, and PC based on breath testing with a novel nanosensor approach. With this in mind, the lab (a) modifies an array of nanosensors for obtaining highly-sensitive detection levels of breath biomarkers for cancer; and (b) investigates the use of the developed array in a clinical study. Towards this end, researchers collect suitable breath samples from patients and healthy controls in a clinical trial and test the feasibility of the device to detect LC, BC, CC, and PC, also in the presence of other diseases. We then investigate ways to identify the stage of the disease, monitor the response to cancer treatment, and identify cancer subtypes. Further, we use the developed device to monitor cancer patients during and after treatment. The chemical nature of the cancer biomarkers will be identified through spectrometry techniques.
The proposed approach can be used outside specialist settings. It could considerably lessen the burden on health budgets, both through the low cost of the proposed all-inclusive cancer test, and through earlier and, hence, more cost-effective cancer treatments.
LCAOS: A Nanoscale Artificial Nose to Easily Detect Volatile Biomarkers at Early Stages of Lung Cancer and Related Genetic Mutations
In LCAOS, the lab aims to enable the earliest possible detection of lung cancer (LC) using volatile biomarkers present in exhaled breath and/or headspace of LC tissues/cells by applying a novel, non-invasive, easy-to-use tool. This tool will be based on an artificial (electronic) nose, which makes use of cross-selective and sensitive sensor arrays trained in their ensemble [R1] to detect LC biomarkers.
In LCAOS LNBD researchers uniquely modify this artificial nose concept by using 3D silicon nanowire- (Si NW) based field effect transistors (FETs) that show increased sensitivity to LC biomarkers. They term this new Nano Artificial NOSE concept ‘NA-NOSE’ throughout the proposal. This ‘NA-NOSE’ will be used to screen and identify high risk groups for LC, and monitor the therapy provided to people with LC to establish point-of-care diagnostics and less invasive treatments. Testing of the novel ‘NA-NOSE’ for early detection of LC, essentially from breath and tissue headspace samples, is conducted by:
(a)Lab-track evaluation – carried out using simulated compositions of clinical samples and, also, real clinical samples, which will be provided by clinical partners.
(b)Clinical-track evaluation of an integrated, professionalized, easy-to-use ‘NA-NOSE’ tool that is carried out at clinical institutes.
The ‘NA-NOSE’ would be suitable for use outside specialist settings and would significantly reduce costs in health budgets. In addition, the ‘NA-NOSE’ could be used for immediate diagnosis of fresh LC tissues in operating rooms, where a dichotomic diagnosis is crucial to guide surgeons. The easy-to-use — no medical specialists required – ‘NA-NOSE’ technology detects cancer based on a change in the blood chemistry and/or metabolic activity. This change is reflected in the chemical composition of the exhaled breath (and cell/tissue headspace) rather than by tumor imaging, thus permitting the earliest cancer detection –. before a tumor of detectable size has formed. The effectiveness of the ‘NA-NOSE’ in detecting LC volatile biomarkers specifically and selectively will provide a launching pad for identifying other types of cancer including breast, colon, and prostate cancers from simple analysis of clinical samples. . Biomarkers that indicate other diseases, such as heart failure, kidney disease and liver disease, can be also tested with the same easy-to-use ‘NA-NOSE’ approach.
i-PEN: Innovative Photonics Education in Nanotechnology

Technion is one of nine partner universities to take part in the iPEN (Innovative Photonics Education in Nanotechnology) has as a primary objective to provide an education training program to young researchers (postgraduate, Research students) in the most common used photonic tools & techniques in a Nanotechnology Laboratory. iPEN project during its development will include the development of online & offline modules, the organization of intensive courses, that will foster the photonic learning skills and build the confidence of young researchers in the field of Nanotechnologies. The project was kicked off in the Oct 2017. iPEN has received a grant as part of the Capacity Building in Higher Education program in the framework of Erasmus+. iPEN project consists from a multinational consortium, with partners from Greece, Italy, Germany, Netherlands, and Israel.
COSMIC: CBRNE Detection in Containers

Technion is one of nine partners to take part in the COSMIC project.The threat of CBRNE (Chemical, Biological, Radiological, Nuclear and Explosives) components used by terrorists is a major concern for EU and worldwide security. Today there is a major security gap in the existing security flow that can be exploited by terrorists to hide and smuggle CBRNE materials inside containers and vehicles. The challenge of improving container and vehicle border crossing and critical infrastructure entrance security checks is of great importance in fighting terrorist threats, theft and smuggling. A study by the EC indicates some of the main CBRNE threats including biological weapons which could be used to attack people in city centers or infrastructure. It was also reported that in the US alone, there were more than 1,300 reported incidents of lost, stolen, or abandoned devices containing sealed radioactive sources. Report summarizes the “historical attacks using chemical or biological weapons” list 23 attacks, demonstrating the attempts and capability of terrorists to acquire CBRNE materials to prepare CBRNE bombs.
VOGAS: Screening of Gastric Cancer via Breath volatile organic compounds by Hybrid Sensing Approach
 Gastric cancer has good chances to be cured if diagnosed at an early stage; currently, there is lack of a reliable non-invasive screening tool. Eastern Europe and Latin America are among the geographical areas with high burden of the disease.
Gastric cancer has good chances to be cured if diagnosed at an early stage; currently, there is lack of a reliable non-invasive screening tool. Eastern Europe and Latin America are among the geographical areas with high burden of the disease.
This project is aiming at developing a non-invasive gastric cancer screening tool by detecting characteristic panels of volatile organic compounds (VOCs). The mechanisms of VOC origin and their composition in gastric cancer patients will be addressed via headspace analysis of cancer tissue, cell-lines, and bacterial flora; VOCs identified during this analysis will be compared to the compounds present in human breath, both via gas chromatography coupled to mass spectrometry (GCMS).
Based on this data, a new type of breath analyser will be developed and validated building upon previously acquired expertise of the consortium members. Specifically, the exhaled breath analyser will combine electronic nose concepts utilizing gold nanoparticles and metal oxide sensors with orthogonal mid-infrared spectroscopic techniques for obtaining multi-dimensional analytical data sets; furthermore, compact pre-concentration schemes will be evaluated. All components will be integrated into several prototypes of portable and optimized devices.
The clinical studies will be conducted in parallel in European (Latvia, Ukraine) and Latin American (Colombia, Brazil, Chile) countries with high disease burden. Patients with gastric cancer and individuals with and without precancerous lesions (dysplasia, atrophy, intestinal metaplasia) will be enrolled. The role of confounding factors including H.pylori infection will be addressed.
As the main result, we expect to provide the first reliable non-invasive gastric cancer screening device based on exhaled breath analysis concepts for future large-scale implementation. The development of according research competences and international collaborations therefore is the most vital component of this project.
A-PATCH: Autonomous Patch for Real-Time Detection of Infectious Disease

A-Patch aims to research, develop, and validate a novel non-invasive wearable sensing patch for detection of infectious disease at point-of-care, such as Tuberculosis (TB), from the skin, with an ability to serve as a monitoring and epidemic control tool.
The device will be a wearable autonomous sensing patch incorporating newly printed sensors and thin-film oxide based flexible electronics, self-repairing components, self-powered components, and a communication layer, for wireless transfer of sensor readout. The patches will be produced in a pilot-line setting for validation trials, and at the same time aligned for subsequent volume manufacturing. A supply-chain and roadmap for large-scale production, regulatory approvals, and go-to-market strategy, will be prepared to enable full post-project commercialization and exploitation of the A-Patch platform.
ONCOSCREEN: A European “shield” against colorectal cancer based on novel, more precise and affordable risk-based screening methods and viable policy pathways

Modern healthcare systems need to offer new non-invasive methods for colorectal cancer screening and to improve its early detection. Such methods need to be affordable, accessible, and applicable to large parts of the population in order to effectively prevent the development of colorectal cancer.
ONCOSCREEN project responds to this challenge and plans to develop a ground-breaking set of technologies and methods for colorectal cancer screening. In close cooperation with other Mission Cancer projects, ONCOSCREEN will provide solutions for risk-stratified cancer screening programmes for citizens, an integrated diagnostic decision support tool for clinicians as well as intelligent monitoring tools for policy makers. The project will collaborate closely with national cancer mission hubs to facilitate policy dialogue on cancer and related research actions.
The generated ONCOSCREEN tools will be extensively validated through clinical studies with 4100 European citizens and patients participating from 10 different countries. The views and perspectives of citizens and patients will be incorporated through a participatory co-design approach, further reinforced by open innovation and FAIR data. The aim is ultimately to have the solutions adopted by public healthcare systems and everyday clinical practice across Europe.
LUCIA: Lung Cancer-Related Risk Factors and Their Impact Assessment

LUCIA is a project funded by the European Union Horizon Europe Programme. It started in January 2023 and will last 48 months. It brings together a consortium of 20 partners – academia, associations, hospitals, NGOs, research and technological organisations, and industry – led by TECHNION (Israel Institute of Technology), from six EU countries and three associated countries. Based on a multi-stakeholder approach, their expertise covers all parts of the process: technology provider, clinical validation, biological understanding, ethics and legal management, policy making, communication and dissemination.
Lung Cancer (LC) is the cancer killing the most worldwide. Though tobacco smoking has long been recognised as the major risk factor, many cases cannot be explained by this reason. In addition, Low-Dosed Computed Tomography, the most common method of LC screening, has remained very little used, with low reduction of lung cancer mortality. In this context, the key aim of LUCIA is to constitute a toolbox for discovering and understanding new risk factors that contribute to LC development.
MELCAYA: Novel Health Care Strategies for Melanoma in Children, Adolescents and Young Adults

In childhood, adolescence and young adults (CAYA), melanoma is under-studied and non-existing tailored clinical guidelines and standardized approaches lead to a very low diagnostic accuracy. Against this background, the MELCAYA project aims to understand risk factors and determinants of melanoma to improve the prevention, diagnosis and prognosis of melanomas in CAYA. To achieve these goals, the project will implement the following approaches.1) By integrating existing reference European cohorts and registries, studies of genetic and environmental risk factors and progression of melanoma in CAYA will be performed through different omic methods, and a novel taxonomy of CAYA melanoma will be generated. 2) MELCAYA will develop image-based robust and trustworthy machine learning tools and a pan-European second-opinion platform for better diagnosis specifically designed for CAYA. 3) Moreover, the validation of minimally and non-invasive disruptive tools based on artificial intelligence and volatilomics detection from exhaled breath and skin will lead to earlier detection and more accurate prognosis of melanoma in CAYA. 4) Finally, through the evidence gathered, MELCAYA will design and implement public health strategies and will actively involve patients and the general population. The results of MELCAYA will maximize its impact by making its data and results accessible and re-usable through integration into UNCAN.eu.
VOLABIOS: Validation and Comparitive Multi-Omics Benchmarking of Fluid-Derived Volatilomics Biomarkers for the Prevention and Early detection of Schizophrenia

VOLABIOS is a groundbreaking European research project under the Horizon Europe framework, dedicated to transforming the way schizophrenia is diagnosed and monitored. Combining cutting-edge technologies, interdisciplinary collaboration, and patient-centered solutions, VOLABIOS aims to address the global challenge of diagnosing and managing this complex mental health condition.
We strive for a future where schizophrenia is detected early, diagnosed accurately, and treated effectively, enhancing the quality of life for millions worldwide. Through innovation, we develop advanced, non-invasive diagnostic tools to improve precision and reduce errors.
By fostering collaboration among experts from 18 organizations across eleven European countries, VOLABIOS is not only advancing early schizophrenia (SZ) diagnostics but also delivering a suite of impactful solutions. These include cutting-edge portable technologies for real-time diagnosis using VOC biomarkers, a Clinical Decision Support System (CDSS), and a mobile health application tailored to patients and clinicians.